News
-

Arc Furnace Graphite Electrode Manufacturer
Graphite electrodes are integral components in the functioning of arc furnaces, playing a vital role in numerous industrial processes. 1. Introduction to Graphite Electrodes: Graphite electrodes are conductive rods made from graphite materials. They function as conductors of electric curr...Read more -

Electric Arc Furnace Graphite Electrode Manufacturer
Graphite electrodes are crucial components in the operation of arc furnaces, serving a vital role in various industrial processes. Graphite electrodes are primarily made from a form of carbon called graphite, which is a crystalline form of the element carbon. Graphite possesses unique properties...Read more -

Graphite Electrodes are Connected by Nipples
In the steel industry, where efficiency and productivity are paramount, the use of graphite electrode nipples has become an indispensable practice. These nipple connectors facilitate the transfer of electric current and maintain a stable arc in electric arc furnaces, which are widely employed for...Read more -

Solutions to reduce graphite electrode consumption
Graphite electrodes are an essential component in various industrial applications, particularly in the steel manufacturing sector. These electrodes play a crucial role in electric arc furnaces, where they are used to create the high temperatures needed for melting and refining metals. However, th...Read more -

Graphite electrodes used for electric arc furnace in steelmaking
Graphite electrodes play a vital role in the steelmaking process, specifically in electric arc furnaces. These high-quality graphite electrodes are designed to withstand large electrical currents and extreme temperatures, making them essential for efficient and effective steel production. When ...Read more -

Multiple factors affecting graphite electrodes price
Graphite electrodes play a crucial role in various industries, particularly in electric arc furnaces. These electrodes conduct electricity and generate intense heat, necessary for melting and refining metals. As a result, they are essential for steel production, scrap metal recycling, and other m...Read more -

Use of Electrode Paste
Electrode Paste, also known as Anode Paste, Self-baking Electrodes Paste, or Electrode Carbon Paste, is an essential component used in various industries including steel, aluminum, and ferroalloy manufacturing. This versatile substance is derived from a combination of calcined petroleum coke, cal...Read more -

What is a silicon carbide crucible used for?
Silicon Carbide (SiC) Crucibles are premium-quality melting crucibles designed to provide exceptional performance in various industrial applications. These crucibles are specifically engineered to withstand high temperatures of up to 1600°C (3000°F), making them ideal for melting and refining pre...Read more -

What are uses for graphite electrode
Graphite electrodes, often referred to as graphite rods, play a crucial role in various industries due to graphite electrodes properties and versatile applications. I:Graphite electrodes are primarily used in electric arc furnaces (EAFs) for steel production. EAFs are increasingly replacing trad...Read more -

Graphite Properties-Thermal Conductivity
Graphite is a unique and exceptional material that possesses remarkable thermal conductivity properties.The thermal conductivity of graphite increases with the increase of temperature, and its thermal conductivity can reach 1500-2000 W / (mK) at room temperature, which is about 5 times that of co...Read more -

Why Graphite Electrodes Are Used in Electrolysis?
Electrolysis is a technique that uses an electric current to drive a non-spontaneous chemical reaction. It involves the splitting of compound molecules into their constituent ions or elements using the process of oxidation and reduction. Graphite electrodes play a crucial role in facilitating ele...Read more -
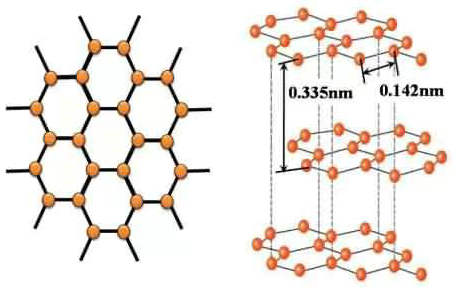
What is the chemical formula for graphite?
Graphite, molecular formula: C, molecular weight: 12.01, is a form of element carbon, each carbon atom is connected by three other carbon atoms (arranged in honeycomb hexagons) to form a covalent molecule. Because each carbon atom emits an electron, those that can move freely, so graphite is a co...Read more


















